Researchers have detailed the inner workings of a critical bacterial enzyme, the Na+-NQR pump, revealing a precise 'dual-trigger' mechanism involving sodium binding and electron transfer that drives its function. This insight into how pathogenic bacteria generate energy opens avenues for developing novel antibiotic treatments.
The Na+-pumping NADH-quinone oxidoreductase (Na+-NQR) enzyme is essential for the energy production in numerous disease-causing microbes, including the bacterium responsible for cholera. This molecular machine moves sodium ions out of the bacterial cell, establishing an energy gradient crucial for functions like motility and nutrient absorption. A significant challenge in understanding this enzyme has been visualizing its dynamic, transient movements as it operates.

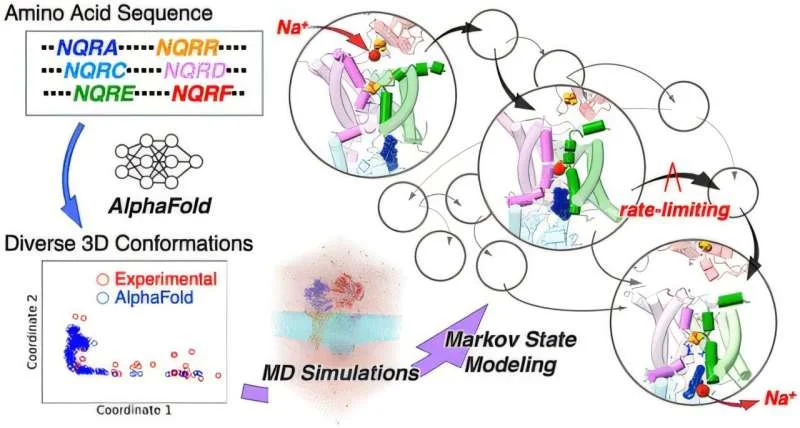
New research, utilizing advanced computational simulations aided by modified artificial intelligence techniques, has successfully mapped the enzyme's step-by-step operation. The simulations provided a clear picture of the energy requirements and the specific pathways taken by the enzyme's subunits, NqrD and NqrE, during sodium transport.
Read More: New Light-Matter Particles Could Speed Up AI Computers

Unraveling a Biological Puzzle
For years, scientists have grappled with the exact link between electron transfer processes, which involve the exchange of electrons between molecules, and the subsequent movement of sodium ions across the cell membrane. This enzyme, unlike many human energy-converting systems that use proton gradients, specifically employs sodium ions.

The detailed analysis has shown that the binding of sodium ions and the transfer of electrons act in concert. This 'dual trigger' precisely controls a cycle of shape changes within the enzyme's subunits, facilitating the pumping of sodium ions. This understanding resolves a long-standing mystery in bacterial bioenergetics, providing unprecedented clarity on how complex molecular machines achieve ion transport.
Implications for Antibiotic Development
Given the Na+-NQR enzyme's vital role in the survival and function of pathogenic bacteria, it represents a prime target for the development of new antibiotics. By understanding the precise mechanics of its operation, researchers can now explore ways to selectively inhibit its activity. Future investigations are planned to determine if the identified structural states of the enzyme can be targeted to block its operation.
Read More: Bangor church syringe program causes litter, hurts business
Background: Bacterial Energetics
Pathogenic bacteria harness energy through various mechanisms. While some utilize proton gradients, the Na+-NQR system relies on sodium ions. This ion gradient is used to power essential cellular activities. The lack of detailed structural information on the intermediate states of the enzyme during its operation has historically hampered scientific understanding. Previous work has explored the general roles of efflux pump proteins in antibiotic resistance, but the specific mechanism of the Na+-NQR pump was less understood.