Scientists have discovered a previously unknown "pocket" on a protein called cereblon, a crucial element in the development of billion-dollar drugs. This revelation shifts the focus from cereblon's main interaction point, long associated with the drug thalidomide, to a secondary, "allosteric" site. This new understanding could pave the way for enhanced cancer therapies, potentially boosting drug effectiveness and reducing harmful side effects.
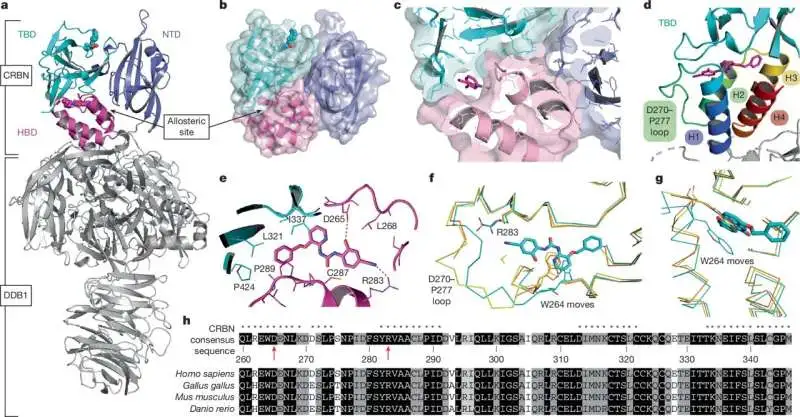
The research highlights that this secondary pocket does not replace cereblon's primary binding site. Instead, it alters the protein's behavior after being engaged. This mechanism offers a new avenue for drug design. One proposed application involves using molecules that bind to this new pocket as adjuncts to existing cereblon-based cancer medications. Such additions could amplify the drugs' intended impact on specific cellular targets while simultaneously mitigating the breakdown of proteins linked to adverse reactions.
Read More: NHS medical history gap in UK 2024 makes health diagnosis hard for adopted children
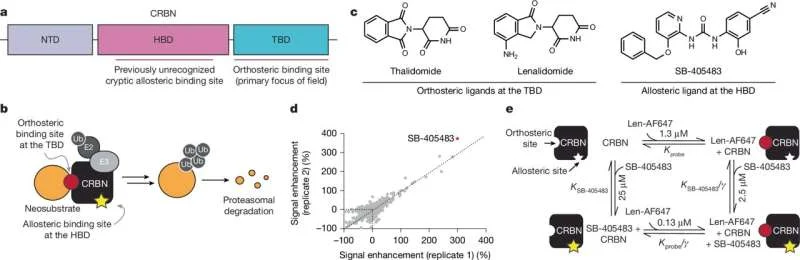
However, the effectiveness of these "allosteric ligands" appears to be dependent on the specific cereblon-binding drug they are combined with. Early findings, including work by GSK scientists who identified a molecule named SB‑405483, suggest this molecule influences certain cellular signals, indicating it might interact with cereblon at a location distinct from its primary site.
Beyond direct therapeutic applications, this discovery offers a glimpse into the intrinsic ways cells might regulate cereblon and similar enzymes. The identification of this allosteric site challenges previous assumptions about how cereblon functions and opens up broader questions about cellular regulation mechanisms.
Background
Cereblon is a protein central to the mechanism of action for a class of drugs, including thalidomide and its derivatives. These drugs have found significant application in treating certain cancers and other conditions. Historically, research and drug development efforts have concentrated on the "orthosteric" binding site of cereblon, the location where thalidomide directly attaches. The recent findings, published in outlets like the Harvard Gazette and Phys.org, introduce the concept of an "allosteric" site – a secondary location on the protein that, when bound by a molecule, influences the protein's overall shape and function. This distinction is critical for understanding how to modulate cereblon's activity in new and potentially more refined ways.
Read More: New 5-Minute Water Test Detects PFAS Forever Chemicals in USA During May 2024


