OFFICIALS EYE UNSEEN CONSEQUENCES AMIDST ESCALATING USAGE
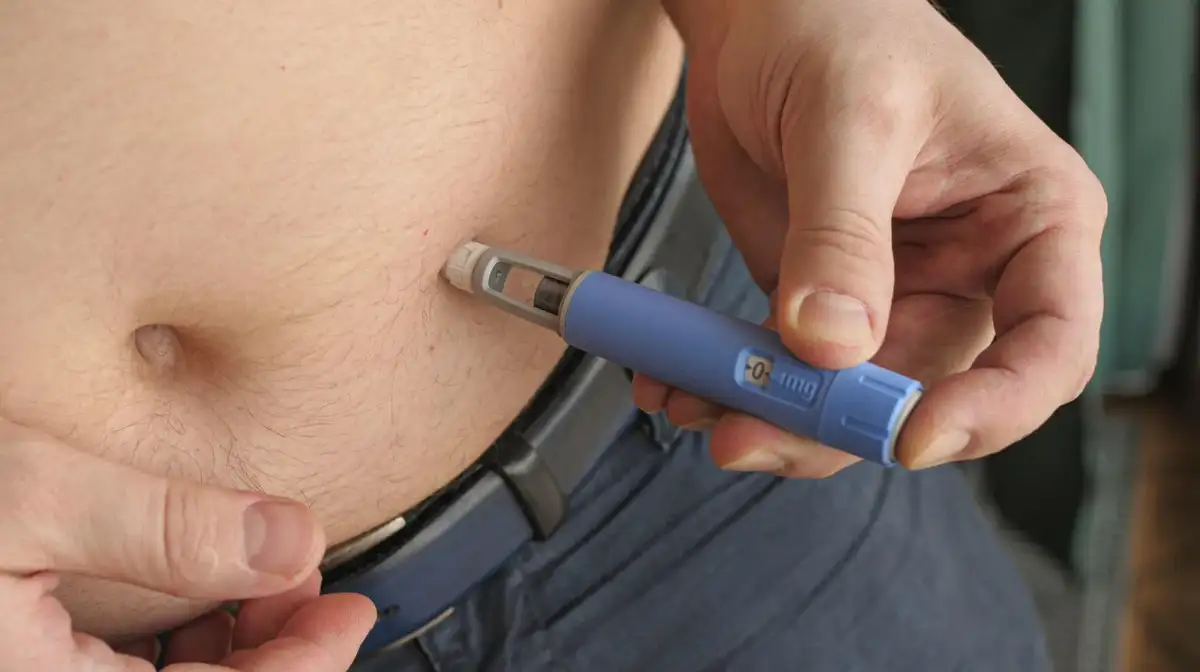
Federal regulators are probing the accumulating reports of serious adverse effects linked to Ozempic and similar drugs, a wave of scrutiny emerging as the medications, primarily designed for Type 2 diabetes, are increasingly adopted for weight management. A confluence of reports, some surfacing mere days ago, highlights a growing concern over adverse events, including severe gastrointestinal issues such as gastroparesis and bowel obstruction. Studies suggest a notably higher incidence of these conditions among users of GLP-1 drugs like semaglutide (the active ingredient in Ozempic) when contrasted with alternative treatments.

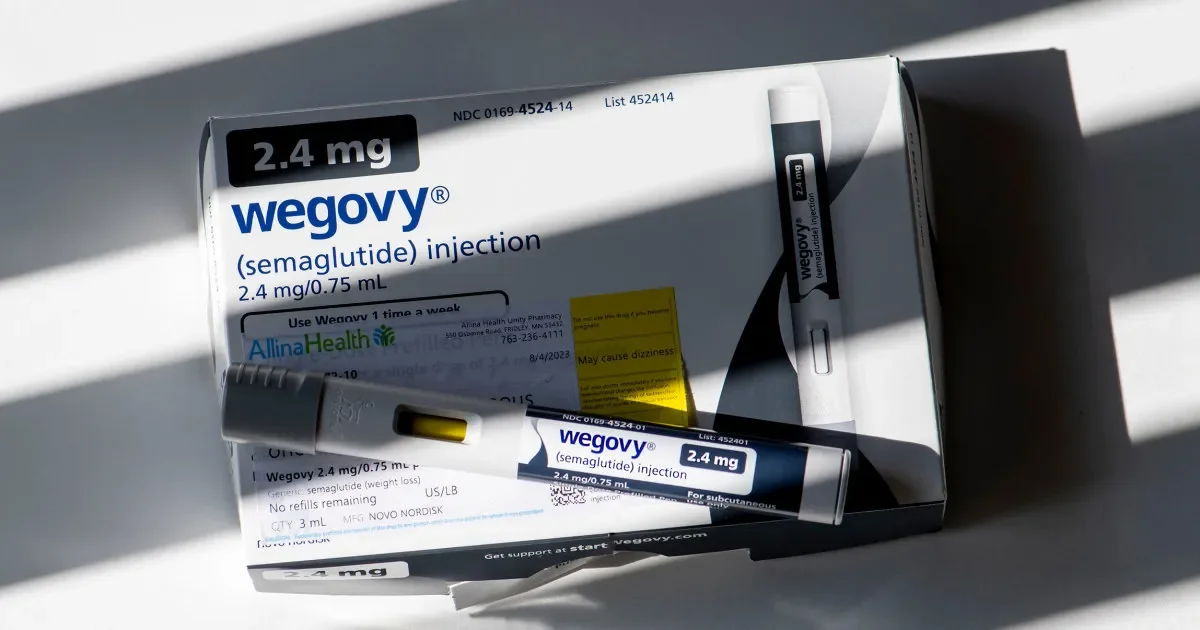
While some research indicates that serious side effects from semaglutide are uncommon, a significant number of users and medical professionals have voiced apprehension about unforeseen dangers. These apprehensions are fueling legal actions, with lawsuits alleging that Ozempic and its related counterparts, such as Wegovy, have led to conditions like NAION (non-arteritic anterior ischemic optic neuropathy). The manufacturer, Novo Nordisk, has acknowledged that product labels for Wegovy and Saxenda do include warnings for potential issues like pancreatitis and delayed gastric emptying, underscoring a disconnect between prescribed use and reported outcomes.
Read More: Zoe Ball's Father Johnny Ball Recovers from Prostate Cancer at 87
A COMPLEX REALITY BEYOND THE NUMBERS
The discourse surrounding Ozempic is multifaceted, grappling with both its therapeutic applications and its escalating off-label use for weight reduction. Initial clinical trials, conducted prior to its FDA approval, identified common side effects. However, post-approval, a subset of patients and physicians have flagged more significant, and sometimes severe, reactions. Concerns have also surfaced regarding the potential for weight regain after discontinuing the medication, suggesting limitations in its long-term efficacy for persistent obesity.
Efforts to manage weight through pharmaceutical interventions are not without their complexities. Some experts are advocating for alternative approaches, such as plant-based dietary interventions, which have reportedly shown promise not only in weight loss but also in improving broader health indicators.

RISKS OF OVERDOSE AND MISCALIBRATION
Reports are also emerging about individuals taking more than the prescribed dosage of semaglutide-based drugs, a situation that can escalate adverse effects to a point requiring hospitalization. Medical guidance firmly advises against any adjustments to Ozempic dosage without direct consultation with a healthcare provider, emphasizing that self-modification carries significant risks, including the potential for permanent damage.
Read More: Senate Republicans Ask Democrats About Voter ID Support After SAVE America Act Fails
The landscape of Ozempic's reception is marked by an increasing number of reported side effects:
Allergic Reactions: Users have documented instances of allergic responses to the medication.
Gallstones: Though reported rarely, gallstones have been identified as a potential adverse event.
Kidney Issues: Some patients have experienced problems related to kidney function.
BACKGROUND TO THE DEBATE
Ozempic, the brand name for semaglutide, was initially sanctioned for the management of blood sugar levels in individuals diagnosed with Type 2 diabetes. Its efficacy in this area is established. However, the drug's significant impact on appetite and subsequent weight loss has propelled it into the spotlight as a widely sought-after tool for weight management, even among individuals without diabetes. This broadening of its perceived utility has amplified the debate surrounding its benefits versus its potential harms, a discussion now under the watchful eye of regulatory bodies and a growing number of concerned individuals.








