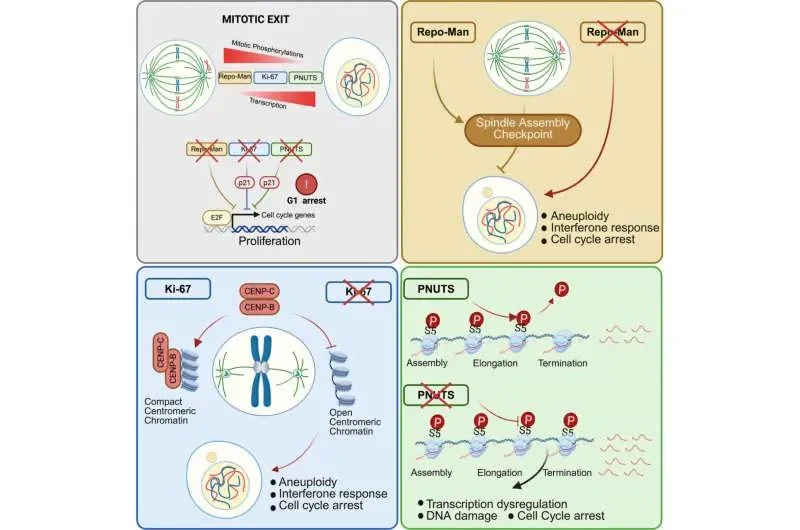
Researchers have pinpointed the protein Ki-67, a ubiquitous component of human cells, as a potential target for cancer therapies. Ki-67 plays a crucial role in maintaining the structural integrity of centromeres, vital regions of chromosomes that ensure accurate cell division. Studies, including work from Brunel University of London, suggest that this protein, alongside Repo-Man and PNUTS, is integral to the complex process of cell division, where the intricate system of gene expression switches is reconfigured for each new cell. The proper functioning of these proteins is critical, as errors in this process can lead to aneuploidy, a condition observed in developmental disorders like Down syndrome and, notably, in many forms of cancer.

Protein Activity and Cellular Identity
The fundamental challenge in cell division lies in the re-establishment of cellular identity. Every human cell shares identical DNA, yet the distinction between a liver cell and a brain cell is determined by which genes are activated and which remain dormant. The study highlights that Ki-67, Repo-Man, and PNUTS are key players in rebuilding the molecular machinery that dictates this cellular programming during division.
Read More: Google Fitbit Adds Gemini AI For Personal Health Coach

Broader Avenues in Cancer Research
Beyond Ki-67, the landscape of cancer therapeutic targets is expanding. A separate investigation published in 'Nature Communications' explored a broader spectrum of 2074 circulating proteins, identifying pathways relevant to cancer development and potential targets for prevention. This research specifically flagged 'EGFR' as a gene linked to breast cancer risk and examined the feasibility of mapping these risk proteins to existing drug targets, while also considering potential adverse effects of interventions.
Meanwhile, researchers at Johns Hopkins Kimmel Cancer Center have uncovered a different angle: a novel tumor-suppressive mechanism. Their work focused on inhibiting 'RNA Polymerase 1' (Pol 1), an enzyme central to the production of ribosomal RNA. This inhibition triggered a distinct cellular stress response that, in turn, altered how cancer cells process RNA, ultimately leading to the suppression of tumor growth. This process, involving the rewiring of splicing—the way cells create different protein forms—may also influence how the immune system recognizes tumors. A small molecule, 'BMH-21', developed by Johns Hopkins pharmacologists, was utilized in these studies.
Read More: Water fleas use IR25a and IR93a receptors to grow protective helmets