Coordinated Motion Unlocks Potential for Precision Therapies
Scientists have elucidated a key principle in the movement of microscopic entities: their speed and effectiveness amplify when they operate in concert. This discovery, emerging from sophisticated computer simulations and theoretical frameworks, offers a tangible pathway toward developing "mesoscale" or microscopic robots capable of navigating the human body for targeted drug delivery.
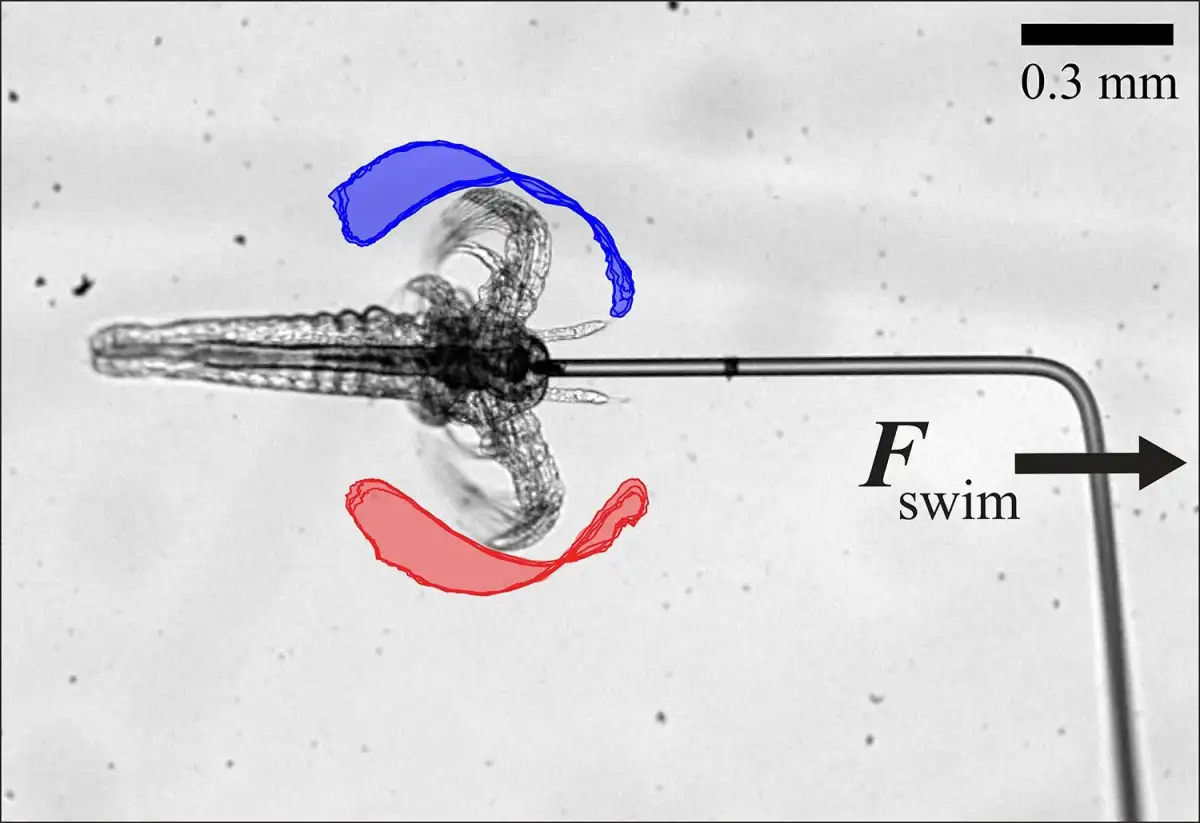
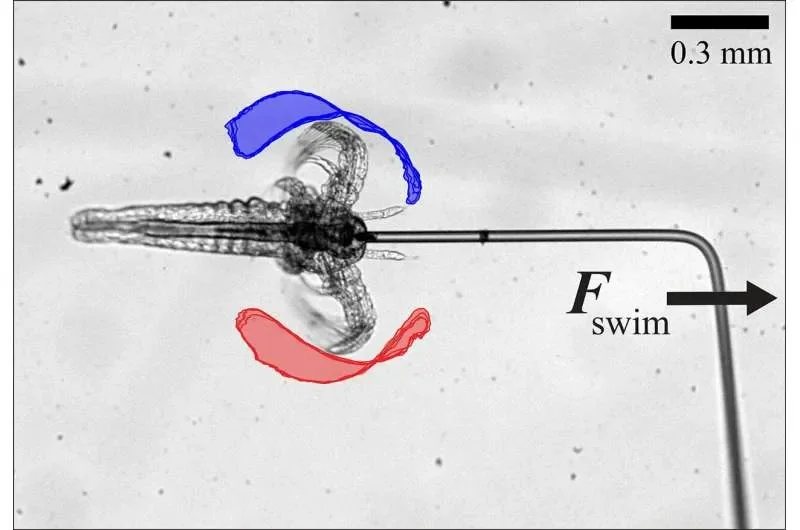
The core finding hinges on the observed phenomenon that as the number of individual microswimmers increases, their collective average speed also escalates, surpassing their solitary capabilities. Furthermore, the study highlights the influence of specific fluid environments, namely liquid crystals, in guiding and directing these tiny agents. These unique fluids, characterized by their liquid-like flow coupled with ordered molecular alignment, appear to provide a crucial structural element for controlled movement.
Navigating the Body's Complexities
The implications for medical science are substantial. The research suggests that these artificially designed microswimmers could function as highly precise delivery systems. - They could be engineered to automatically locate and linger over diseased or damaged tissues, which often emit distinct chemical signals. - This targeted approach promises to deliver medication directly to the affected areas, potentially circumventing the systemic side effects associated with traditional drug administration methods.
Read More: New Navigational Mind Theory Links Time and Space Processing
Elongated particle designs, observed to be more stable than spherical counterparts which tend to disperse, are proving more effective for maintaining positional integrity. This stability is paramount for ensuring that drug carriers remain fixed at their intended therapeutic sites. The research signifies a considerable advance in the quest for autonomous microscopic devices, opening avenues for both medical and industrial applications requiring intricate internal manipulation.
Beyond Simulation: The Road Ahead
While current investigations have largely relied on small-scale simulations, the next phase aims to scale these models up. The objective is to replicate the movement of hundreds, and eventually thousands, of microswimmers within more complex, enclosed fluid environments that mimic physiological conditions. This expansion of research will be vital for understanding how these entities interact and function within the intricate internal landscape of the body.
Read More: Mounjaro patients stop drinking alcohol suddenly
The foundational understanding of how naturally occurring microswimmers navigate diverse fluid settings is seen as a prerequisite for this advancement. The current study represents significant progress in this regard, offering insights into the fundamental mechanics that could govern the future of internal robotics.
Origins of the Research
This line of inquiry draws inspiration from observations of microscopic organisms and is being explored through distinct, yet converging, research efforts. Studies, such as those published in 'Soft Matter', have focused on Janus particles, a specific type of micro-robot designed to respond to chemical gradients, much like bacteria seeking sustenance. These particles are being investigated for their ability to autonomously detect and orient themselves towards chemical signals emanating from targeted tissues. The collective work underscores a growing interest in harnessing the power of microscopic coordination for complex tasks.
Read More: Why Sudoku puzzles caused seizures in a 25-year-old German man









