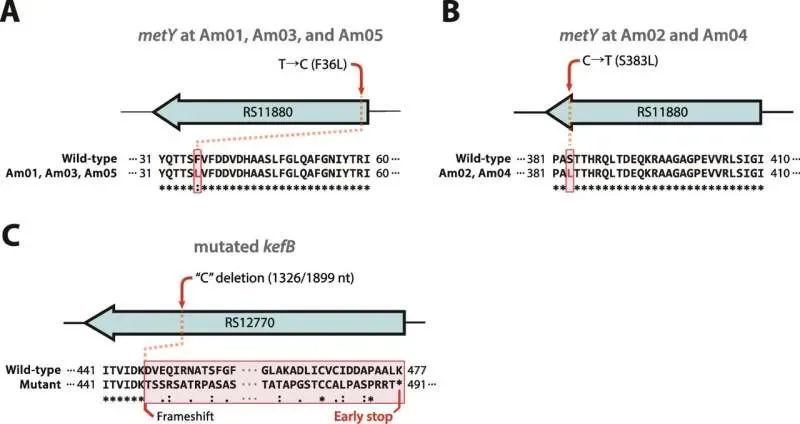
A new DNA barcoding technique has emerged, offering a way to track the fate of nanoparticles designed to deliver gene therapies inside living organisms. This method promises to clarify why many of these therapeutic delivery systems fail to reach their intended targets within cells, ultimately paving the way for more effective treatments.
The core challenge lies in how cells handle foreign material. Upon entering a cell, gene therapy carriers are often rerouted to lysosomes, the cell's internal recycling and waste disposal units. Here, the valuable therapeutic genetic material can be degraded before it has a chance to perform its intended function. For gene therapies to work, these carriers must evade this cellular 'trash system' and arrive at the specific cellular locations where they can be active.
This new assay, developed by researchers at Oregon State University and collaborators, allows scientists to directly measure in vivo how much genetic cargo escapes cellular degradation and how much is destroyed. This capability represents a significant advancement, enabling, for the first time, rapid quantification of nanoparticle performance within living systems. The findings from these measurements have already guided the design of improved lipid nanoparticles. These next-generation carriers incorporate enhanced 'ionizable lipids,' demonstrating the potential for gene editing at substantially lower doses compared to existing approaches.
Read More: Perth Lab Makes DNA for New Medicines, Beyond Weight Loss Drugs
The research, detailed in a publication within Nature Biotechnology, underscores the significance of Gaurav Sahay's work in drug delivery science. Jozić, working under Sahay's direction, spearheaded the practical application of this barcoding method. The study's collaborative nature is highlighted by contributions from institutions across the globe, including Oregon Health & Science University, Tennessee Technological University, Yeungnam University in South Korea, and the University of Brest in France.
Read More: Rudy Giuliani Leaves ICU After Pneumonia, Continues Hospital Recovery
Looking ahead, the insights gained from mapping these intracellular pathways could extend beyond current gene therapies. The technique holds promise for optimizing the delivery of a variety of therapeutic payloads, such as mRNA vaccines, small interfering RNAs (siRNA), and CRISPR-based gene editing tools. This development positions Oregon State University as a notable entity in the fields of drug delivery and biotechnological innovation, with the potential to unlock higher efficacies in gene delivery by strategically navigating cellular defense mechanisms.